|
Hey there! It's Jocelyn. Sorry we are so late getting back to you guys about your comments! Thank you for the compliments! They were very encouraging when we hit rough roads in the middle of research. Now I hope to be able to answer most of your questions.
Q. I am wondering why you chose 5 ml. of debris. Since you are using a smaller solar panel wouldn't you want to scale the average amount of debris on a larger solar panel to your test solar panels? If no data has been created for the average amount of debris on medium sized solar panel, do you think it is possible to find a place that would collect this information for you, perhaps the next time that they clean their solar panels? A. The reason we chose 5 ml of debris was because no one has measured the amount of dust on a solar panel, at least, no one posted it online for us to find. Most people just wash the debris off without measuring how much is on it, because it is nearly impossible. If it was possible, yes, we would have scaled the amount of debris down, but we had to work with what we had. Yes there are many places that could have collected the information for us, but those places are located all over the country, and have no time to do such a thing for us. We would have asked friends that have solar panels, but Michigan does not have as much debris floating in the air as an arid, or oceanic land and therefore they could not be used in this research project. Q. In the blog you said that you had people in mind that you planned to contact and I was curious as to who you were thinking of. A. We did contact a physics professor at a university, however, he never responded to our e-mails. In the end, our professional contact was my uncle, Wesley Anderson, who majored in Physics, and Engineering. Q. Would this experiment turn out differently if there was a different amount of sunlight hitting the solar panels? A. Yes this experiment would have turned out differently if there was a different amount of sunlight hitting the solar panels. We would have gotten lower readings from the voltmeter which would make it harder to tell which type of debris had a greater impact on the results. The amount of light needs to stay constant throughout the trials or else the voltmeter would show a lot of variation and the data would be unable to be compared to each other because not all of the supposed constants were kept constant. The purpose of this experiment was not to see what would happen if the light changed, just what would happen when the debris type changed. Q. I was wondering was there a reason why you decided to use the uni-solar solar panels for your experiment, and why? Do you think different makes/models of panels would give different results? A. The reason we used uni-solar solar panels for this experiment was because they were available to us and we did not have to go out and buy one. Building one would take too much time and carries the risk of setting it up wrong. Yes, I think that different makes/ models would give different results but the purpose of this experiment was not to record how many volts this specific panel would generate. Q. Are you also testing to see what would happen if there were more than 1 of these different dust particles in the air or on the panel? A. No, in this research project we are not doing that, however we do suggest doing that in the conclusion for future research topic ideas. It is a good idea to do that because it is very unlikely that there is just a single type of debris in the air, or on a solar panel, at a given time. Q. There were a couple suggestions to come up with an efficient way to clean the solar panel. A. The most efficient way to clean the solar panel is to simply wipe it off using a warm towel. Since we are trying to conserve money and clean the panel, this is the simplest way. The towel needs to be warm because a cold towel on a warm or hot solar panel could cause the panel to crack. Hand cleaning it ensures we get all the dust, and to double check that, the voltmeter read at about the same starting values every time we cleaned it. Q. Why was this topic originally not appealing? Has this ever been researched before? A. Originally, we wanted to test lithium batteries to see if "overcharging" them could lead to a decrease in battery life. Compared to that, putting dirt on solar panels did not seem as interesting, but our original topic had already been researched by many people. Neither Matt or I found this exact experiment online before. While many people have researched similar things, they were not looking for what we were looking for. The purpose of this experiment was to compare the effects different types of debris have on the voltage output of photovoltaic solar panels. Q. There were many questions as to how we came up with this idea, or the procedure. A. We actually were not the ones to come up with this idea. An MMSTC alumni helped us out when we were struggling to come up with one. One of his friends had suggested it as a future research topic at the end of his research project. We decided to use it because all five of our ideas were rejected at the beginning of the year. Eventually, we learned to love the topic. As for how we came up with the procedure, there was a lot of trial and error, but thanks to our physics teacher and professional contact we were able to come up with a plausible idea that was verified during pre-trials. Thank you to all who commented and asked questions! Your criticism was very helpful and allowed us to hammer out some kinks before we ran into them during trials! The entire experiment will be posted shortly. Thank you for reading and following our research! Matt and I wish you all the best!
0 Comments
Matt here again to talk about our conclusion and results of our experiment.
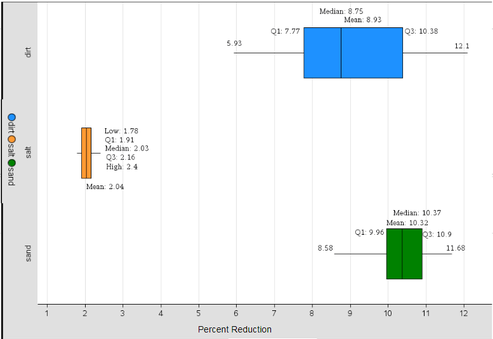
Using the results from the statistical test, it was determined that the sand had the greatest effect on the voltage output of the solar panel. Additionally, the salt solution was shown to have the least effect on the voltage output, meaning our original hypothesis, that sand would have the greatest effect on the voltage output of the solar panel, and the salt solution would have the least effect, is accepted. While one might think the data collected from the dirt may have had the greatest effect, as dirt absorbs the most light of the three debris types used and then re-emits it, the data collected from trials using sand as the debris type had a higher mean than the mean power reduction of the dirt. Additionally, while sand does not absorb as much light as dirt does, sand is able to re-emit and reflect light, therefore preventing more light from reaching the solar panel to be used as power than the dirt. The results of this experiment suggest that installing a solar panel in an area surrounded by sand would not be ideal, as the sand would then cause the most power reduction of the solar panel compared to other regions. Even so, one should expect some voltage reduction regardless of where the solar panel is installed, due to the numerous aerosols (types of debris particles) in the air. Debris may pile on, and begin to reduce the solar panel’s usefulness if not cleaned correctly. Hi everyone, it's Matt today! Here to give you the results of our research, as supported by statistical evidence. Let's get right into it. Last week we took the data we collected and used a two-sample t test to determine which of the three debris types had the most significant effect on the voltage output of the solar panel. The salt solution trials were shown to be much lower than those of the other two, so we did not have to use the data collected from the salt solution trials in our statistical test. To perform a two-sample t test, three assumptions had to be met. These are; the sample population must be a simple random sample, each of the population samples follows a normal probability distribution, and the sample population standard deviations are equal. The first two assumptions were met, but the standard deviations of the two populations being tested (dirt and sand) were not equal. However, each sample population had 30 data points, or trials. Thus, due to the Central Limit Theorem (which states that the assumption of equal standard deviations can be met so long as each sample population has 30 or more data points), we were able to perform the two-sample t test. The null hypothesis for this test stated that the true means of each population were equal, while the alternative hypothesis being that the true population mean of sand was larger than that of the dirt. The test results showed a p-value of 0.00011, a value far less than the alpha level value of 0.05. This means that we reject the null hypothesis, and that there is evidence the true population mean of the sand is greater than the true population mean of the dirt. If the null hypothesis were true, the results obtained in this experiment would only occur about .011% of the time by chance alone. The boxplot above shows the data collected in this experiment. As can be seen, the salt solution had a much lesser effect on the voltage output than the dirt and sand did. Thus, it was decided to perform a two-sample t test on only the sand and dirt. The mean of the sand data is greater than the mean of the dirt data, and the t test concluded the same.
Thanks for reading, and if you have any questions or comments, please let us know in the comments below! Thank you all for the comments and questions! I will try to answer them all in the next post. For now, please comment below on what you thought about our trials!
After pre-trials we went back to the dark room to take pictures however, when we arrived, all the materials for the experiment were missing and we ran all over looking for them. We found the important ones however, after that scare, we were not going to leave the materials in the room so we went back to moving the materials from a locker to the room. This past week Matt and I did the trials for the experiment. We had a two hour block each day from Monday to Friday to get the 90 trials done. We started out strong on Monday and Tuesday by completing 60 of the 90 trials. We alternated between testing the sand and the soil. We had some problems figuring out how the salt trials would be run, which is why we neglected to randomize the trials. On Wednesday we attempted to evaporate the water from a salt solution using a hair dryer as we decided in pre-trials, but decided it would not work. That night, we called our professional contact, Mr. Anderson, and asked him for help creating a salt film on the solar panel. He suggested using spraying water on salt on a clear sheet on saran wrap so it was portable. The microwave was used to evaporate the water from the solution. Many thanks to Mr. Anderson for helping us solve this conundrum. On Thursday we tested 15 of the salt trials and finished the other 15 on Friday. Some observations that were made include the size of the type of debris and the temperature of the solar panel. Over the course of each day, the temperature of the solar panel dropped. This was due to the room temperature of the testing room being lower than the temperature of our lockers. The size of each type of debris was different. The soil contained some rocks, and had the largest granules, the sand had the smallest granules, and the salt had the mid-sized granules. This made distributing each type of debris evenly difficult, especially since there was no method of equal distribution which could have caused some error in the experiment. To view the exact Data and Observations please go to The Experiment tab, and look under data and observations tab. Matt will be typing up the next post about the data analysis. Have a nice week! During this past week, Matt and I started pre-trials. We had a two hour block, every day, from Monday to Thursday to find and resolve any problems that could come up during trials and run some tests. A lot of our time was spent running from the science room to the underground locker room, trying to transport pieces of equipment that we kept in the science room. Later in the week we were given permission to leave the materials in the locker room and that allowed us to focus solely on pre-trials. You will see a slideshow below of pictures and videos we found pertinent to showing how we fixed the initial experimental design. On Monday we gathered all the materials we thought we needed in the locker room and set up a workspace using a couple of desks. We hooked the laser, voltmeter, and solar panel up and turned them on only to find that the laser did not produce enough light for the solar panel to convert to volts for the voltmeter to read. Since no other action could be taken without a light source to start testing the different types of dust, we decided to sift through the sand to get the larger pieces out. On Tuesday we brought in a couple of different flashlights and tested them to see if they would work. However, even when we combined both flashlights, the voltmeter still did not read much higher than the laser. Next we tried a 150 Lumen bulb and our science teacher provided us with a stand that easily allowed us to change the distance between the bulb and the solar panel and it worked! At least, for a while. The voltmeter dropped back down to zero and we could not figure out why. Matt and the science teachers found out that the problem was the metal cords that were hooked up to the solar panel were touching and needed to be moved apart before the voltmeter could read how many volts the solar panel was producing. In the meantime, I dried the dirt out using a hair dryer to save some time. Wednesday we started testing the sand and dirt and determined that 10 milliliters of each type of dust would be sufficient to cover the solar panel. We were going to test the different types of dust at different quantities but determined that it would be pointless because the results on the differences in volts should be the same. Matt then sifted through the dirt to get the larger pieces out. I went home and prepared two salt solutions to test the next day. However, it took me half an hour to prep two solutions so Matt and I started devising a new, faster method to prep the solutions. Thursday we tested the two salt solutions, however, one of them spilled and was unusable and the other was taking too long to dry, even with a hair dryer. So we decided to use only half the solution on the solar panel and recorded the difference in volts the solar panel produced. We also noticed that the temperature of the solar panel around 70 degrees hotter than when we started due to the hair dryer blowing hot air on it. Then we tested 5 milliliters of dirt and sand on the solar panel and determined that 5 milliliters was just enough to cover the solar panel. We ran some more trials, however we forgot to shut off the lights in the hall before doing so and the solar panel was not directly below the light for the first two trials so the data collected is not accurate, but it did give us an idea of what to expect during trials. We never did determine a faster way to collect data from the salt trials, but we did find that table salt could be used for the experiment, not just sea salt. Since sea salt is coarser than table salt, it was hard to dissolve into water resulting in a greater amount of time to prep the solutions. Using regular table salt cut the time to prepare the salt solutions in half. After pre-trials were over we found ourselves with some time on our hands and tested the three solar panels that we planned to use to randomize the experiment, each of the same make and model. One of the solar panels produced around 5 volts, another 6 volts, and the last 7 volts, under the same bulb, the same distance away from the light source. This is too much variation to randomize the solar panels, combine the data, and run an ANOVA test and/or a Two-Sample T-test, so we decided to only use one solar panel in this experiment and randomize the order that we test each trial of dust. We also found that if the distance between the light source and the solar panel is decreased then the solar panel produces more volts because the light is hitting the solar panel directly and not reflecting off of other surfaces. However, so long as the distance between the solar panel and light source is kept constant this should not be a problem. That concluded pre-trials for us. If you have any comments or suggestions that would improve our research please comment below! Thanks for reading! This is a video showing how to prep the sea salt solution. Sorry for the quality of the video. I was filming with one hand and trying to demonstrate with the other. The amount of hot water used does not matter so long as the salt dissolves (that water is hot but you cannot see it in the video). You can use any container you want, I just had small Tupperware jars on hand. This is the video of the sea alt being dried. We sped it up and cut some parts out. It actually took us about 20 minutes to do this.
At first, we didn't want to do this topic at all. When the time came to turn in research proposals we thought we had four or five good ideas that we could pick from. They were all discarded due to comments from teachers or articles found online that told us that topic had already been researched and was proven fact. At that point we were crushed and thoroughly out of ideas. That was when lightning struck a friend of ours who knew of our dilemma. It is thanks to Brian saying that we should do something with dust and solar panels that we were able to fully define the topic of our research.
Throughout the initial stages of the project, we looked up sources that could benefit our research. Many articles were found on Google Scholar, PLOS, or SPIE digital library. Some places only allowed us to view an abstract and other articles wanted money to view the entire document. We avoided those, although there were many promising articles that could have been used. However, the other articles were promising too. An article that detailed how photovoltaic solar panels worked allowed us to know that we were measuring the potential difference of a solar panel. Others told us about similar research that we could use to shape our experimental design. Still another gave us a large hint in the direction we wanted to take the project. This article detailed the dust in the air was actually made up of different things and that was when we decided we wanted to compare how sea salt, dry dirt, and sand, some of the most common forms of dust in the air, affect the potential difference of a photovoltaic solar panel. To date, we have not contacted anyone outside of our teachers about help for this project, although we do have people in mind. Other than making sure the light shines at the same angle on every solar panel, we do not have plans to build anything. Sorry to those who thought we would build our own solar panels. As of right now we have a preliminary experimental design done that allows us to quickly measure the amount of potential difference of the solar panels, while shining a light on them at the same angle every time. All we need to do is put differing amounts of dust on the panel and stat measuring. That's about it for now. Thanks for reading and if you have anything you would like to say please comment below! |